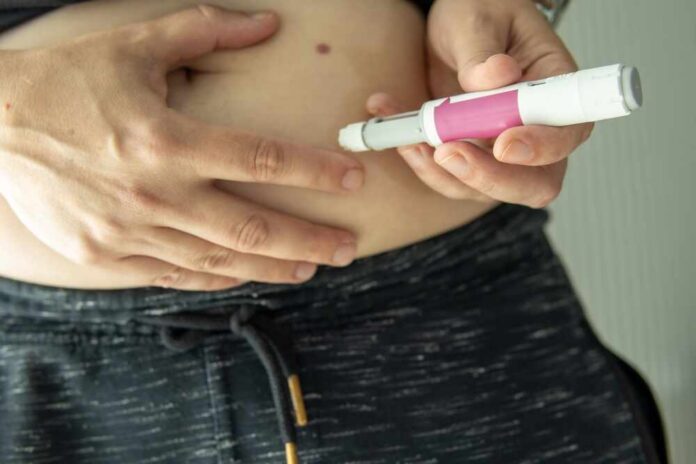
Americans are being told “life-changing” weight-loss injections can prevent heart attacks and strokes—while the hard questions about cost, safety, and government-style mass prescribing still haven’t been answered.
Story Snapshot
- Multiple outlets are highlighting GLP-1 weight-loss drugs as a way to cut cardiovascular risk, not just pounds.
- Supporters call the drugs a breakthrough; skeptics worry the story is racing ahead of long-term data and real-world constraints.
- Any large-scale rollout would collide with familiar problems: high prices, supply limits, and pressure on taxpayers and insurers.
- Conservatives wary of “one-size-fits-all” health policy want clear standards, transparent evidence, and voluntary, informed consent.
What’s Driving the “Heart-Protection” Weight-Loss Drug Push
News coverage and medical headlines have increasingly tied GLP-1 drugs to reduced cardiovascular events, framing them as more than cosmetic weight-loss tools. A central claim is that, in certain patients with obesity and elevated cardiovascular risk, these medications may lower the odds of major heart problems when used consistently and alongside lifestyle changes. That shift in messaging matters because it encourages broader prescribing—potentially moving from niche use to population-scale demand.
The strongest, most verifiable signal is that regulators are now treating this as a cardiovascular-risk story, not merely a weight story. The FDA, for example, has issued a press announcement describing approval of a treatment intended to reduce the risk of serious heart problems specifically for adults with obesity or overweight and established cardiovascular disease. That kind of labeling milestone tends to influence insurance decisions, clinical guidelines, and consumer expectations.
What the Evidence Can—and Cannot—Prove from the Provided Research
Several social clips and segments emphasize “largest benefit ever seen” style language, while others highlight studies suggesting reduced risk of heart attack. Those headlines may reflect encouraging trial outcomes, but does not include the underlying study documents, detailed endpoints, patient criteria, or limitations. Without those materials, readers should treat sweeping claims carefully and insist on specifics: which drug, which population, what absolute risk reduction, and what tradeoffs.
Conservatives are right to ask whether “preventing heart attacks and strokes” is being communicated as a guarantee rather than a statistical reduction in a defined group. A medication can show meaningful benefits in a controlled trial and still face real-world barriers: adherence drop-off, side effects, discontinuation, and unequal access. When the public conversation jumps directly from “promising data” to “million-person prescribing,” the risk is policy by headline instead of policy by proof.
Costs, Coverage, and the Risk of Back-Door Mandates
GLP-1 drugs are widely discussed as expensive, and the provided research set repeatedly frames them as “life-changing,” language that can build political pressure for broad coverage. The problem is straightforward: if the drugs become treated as standard prevention, insurers and public programs face rising costs. For a conservative audience already burned by overspending and inflation, the key question is whether policymakers will shift the bill onto taxpayers or quietly force coverage through regulation.
The information provided does not include pricing tables, budget forecasts, or actuarial impacts, so any claim about total national cost would be speculation. Still, the structural tension is obvious and familiar: broad eligibility plus high per-patient cost equals a push for rationing, prior authorizations, or coercive “guidance” that functions like a mandate. If Americans are going to be encouraged to take powerful drugs long-term, transparency on who pays, who profits, and who decides is not optional.
Medical Choice, Informed Consent, and Trust After Years of Institutional Overreach
After years of politicized public health messaging, many Americans—especially older conservatives—are skeptical of big institutions insisting they “just follow the science” without showing the work. That’s why the details matter: side effects, discontinuation rates, contraindications, and what happens when patients stop therapy.
From a conservative values lens, the standard should be simple: individuals should have access to truthful information and real choice without pressure from employers, insurers, or federal bureaucracies. If these drugs truly reduce serious heart outcomes in defined populations, patients deserve that option. If the benefits are being oversold to justify a mass-prescribing agenda, that undermines informed consent and invites the kind of government overreach Americans have spent a decade fighting.
The next real test will be whether officials pursue voluntary, patient-led adoption—or whether the usual machinery of subsidies, mandates, and administrative pressure tries to make a complex medical decision into a top-down policy program.